Choline acetyltransferase (ChAT) catalyzes the synthesis of the neurotransmitter acetylcholine (ACh) from choline and acetyl-CoA in cholinergic neurons. ChAT serves as a specific marker for cholinergic neurons in both peripheral and central nervous systems. Dysfuntion of cholinergic neurons underlies aspects of clinical symptoms found in neurological and psychiatric disorders such as Alzheimer’s disease, Down and Rett syndromes.
Evidence shows that the enzyme choline-O-acetyltransferase (ChAT) exists in two forms inside cholinergic nerve terminals, a soluble hydrophilic form and the membrane-associated amphiphilic form. As an example, membrane-bound ChAT was shown to be localized as a peripheral protein that is attached to synaptosomal plasma membrane (Gabrielle et al. 2003). Membrane-bound ChAT has served as the feature condition that allows Advanced Targeting Systems to apply their technology through an affinity-purified antibody to ChAT conjugated to saporin, the ribosome-inactivating protein, to specifically target and eliminate those specific cells.
Anti-ChAT-SAP is made with an antibody using a 22-amino acid peptide from porcine ChAT (GLF SSY RLP GHT QDT LVA QKSS). The targeted toxin recognizes porcine ChAT and is expected to cross-react with rat, mouse, and human.
Gabrielle P, Jeana M, Lorenza EC (2003) Cytosolic choline acetyltransferase binds specifically to cholinergic plasma membrane of rat brain synaptosomes to generate membrane-bound enzyme. Neurochem Res 28(3-4):543-549.
Targeted Toxins
Anti-ChAT-SAP eliminates cells expressing ChAT
available individually (IT-42) | available as a kit (KIT-42) including
Saporin (PR-01), Rabbit IgG-SAP (IT-35) and Anti-ChAT affinity-purified (AB-N34AP)
Antibodies
Antibody to ChAT (AB-N34)
affinity-purified (AB-N34AP)
biotin-labeled affinity-purified (AB-N34APBT)
Alexa488-labeled affinity-purified (AB-N34APFLA)
Other Conjugates Targeting the Cholinergic System
192-IgG-SAP (IT-01) eliminates cells expressing p75NTR in rat
ME20.4-SAP (IT-15) eliminates cells expressing p75NTR in mammals
mu p75-SAP (IT-16) eliminates cells expressing p75NTR in mouse
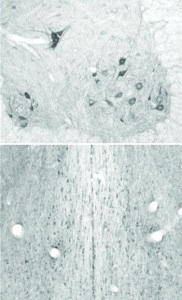
Courtesy of Dr. Ronald G. Wiley. and Robert Kline at Vanderbilt University, Nashville, TN.
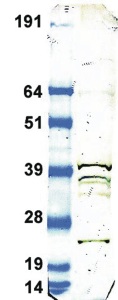
Lane 1: Crude rat brain whole cell extract probed with at 1:100 dilution
Lane 2: Molecular weight standards (Invitrogen SeeBlue).
