Gene therapy has been proposed for a variety of genetic disorders, including severe combined immunodeficiency, but it has not yet found routine use in the clinic, in part because of potential complications. To help pave the way for safer translation of such gene therapy, Schiroli et al. studied potential approaches to gene therapy in mouse models of severe combined immunodeficiency.
The authors analyzed outcomes to find the safest most effective method based on:
- Different approaches to conditioning,
- Different numbers of gene-edited cells,
- Different techniques for editing the faulty gene.
Preclinical Modeling Highlights the Therapeutic Potential of Hematopoietic Stem Cell Gene Editing for Correction of Scid-X1. Schiroli, G, Ferrari, S, Conway, A, et al. (2017). Sci Transl Med, 9 (411):2017/10/13.
Mice were treated with a single dose of CD45 antibodies conjugated to the protein synthesis inhibitor toxin Saporin (CD45-SAP)*, which caused substantial depletion (~70%) of the HSPC compartments and milder depletion of the more mature cell populations and transplanted with 10% wild-type (WT) cells 3 days later.
*CD45-SAP (biotinylated CD45 antibody mixed with Streptavidin-ZAP)
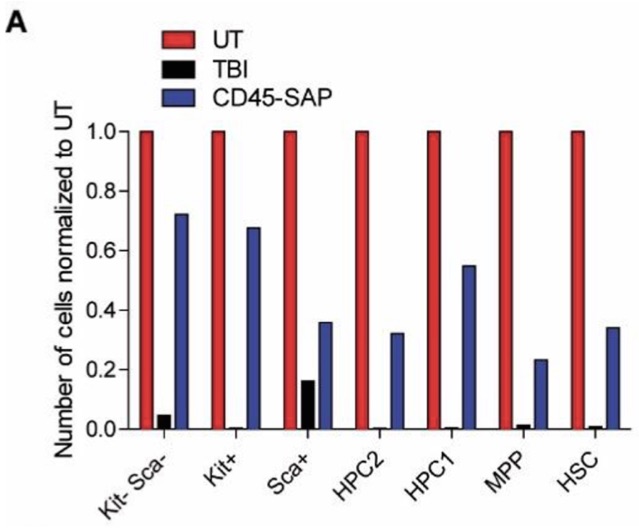
Also see: Non-genotoxic conditioning for hematopoietic stem cell transplantation using a hematopoietic-cell specific internalizing immunotoxin. R. Palchaudhuri et al., Nat. Biotechnol. 34, 738–745 (2016).
