by Ronald G. Wiley, M.D., Ph.D., Departments of Neurology and Pharmacology, Vanderbilt University, Nashville, TN and C. J. Vierck, Department of Neuroscience, McKnight Brain Institute, University of Florida College of Medicine, Gainesville, FL, USA
Degeneration of the cholinergic basal forebrain (CBF: medial septum, diagonal band of Broca, nucleus basalis of Meynert/substantia innominata) is a prominent feature of Alzheimer’s disease (AD). The CBF supplies cholinergic input to most of the cerebral cortex and hippocampus including somatosensory areas and anterior cingulate cortex that are involved in pain perception and experiencing discomfort, respectively. Clinical literature suggests that patients with AD either feel less pain or express discomfort less than comparable patients without dementia. As a result, AD patients receive less analgesics, but there is concern that AD only impairs communicating discomfort. Rats with extensive CBF lesions show impairment in a wide range of learning tasks and ability to sustain selective arousal/attention, but it is not known what role the CBF plays in central pain processing.
The present study sought to assess the impact of CBF lesions on behavioral responses to nociceptive stimuli in rats. Rats were trained on a thermal escape task where they chose whether to spend time in a dark chamber with the floor temperature at 10° C or 44.5° C (both mildly unpleasant), or move to a connected room temperature chamber with bright lighting. After establishing baseline performance on the operant task, selective CBF lesions were produced by intracerebroventricular injection of 192-saporin (192-IgG-SAP, Cat. #IT-01; Fig. 1). This immunotoxin selectively destroys neurons expressing p75NTR, the low affinity neurotrophin receptor that is uniquely expressed by CBF neurons in the forebrain. The rats were retested repeatedly over 19 weeks. On several occasions, the rats were subjected to sound stress prior to escape testing, and the rats were also tested on the thermal plate (hot/cold plate) without an escape option to measure lick guard (reflex) responses with and without preceding stress.
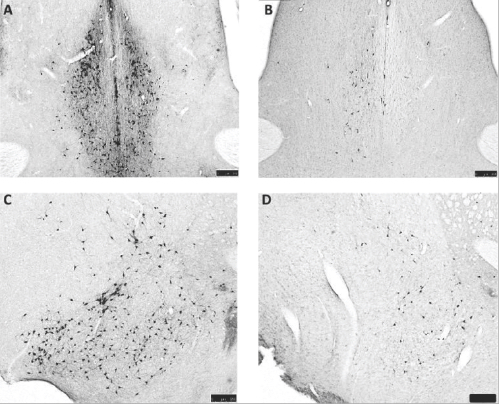
Compared to controls, 192-IgG-SAP injected rats showed highly significant (p<0.001) loss of neurons from all subdivisions of the CBF based on post mortem brain sections stained for choline acetyltransferase. The CBF-lesioned rats escaped less than controls after 192-IgG-SAP injection (i.e. less motivated to get away from the aversively hot or cold stimuli). Reflex lick/guard responses, which are mediated at the spinal level, were not affected. The usual hyperalgesic effect of stress on the operant thermal escape task was absent in the CBF-lesioned rats. These results indicate a role for the CBF in modulating central pain processing. The loss of stress effect on thermal escape responses is consistent with loss of the arousal/attention function(s) of the CBF. These results also demonstrate the usefulness of 192-IgG-SAP for studies of the role of central (cerebral) cholinergic mechanisms in pain processing and are consistent with the idea that AD patients experience less discomfort for a given painful condition.
Reference
- Pain sensitivity following loss of cholinergic basal forebrain (CBF) neurons in the rat. Vierck CJ, Yezierski RP, Wiley RG. Neuroscience 319:23-34, 2016.
