A specific immunotoxin elucidates a causal role of striatal cholinergic system in behavioral flexibility
by Sho Aoki and Jeffery R. Wickens
Neurobiology Research Unit, Okinawa Institute of Science and Technology, 1919-1 Tancha, Onna, Kunigami, Okinawa 904-0495, Japan
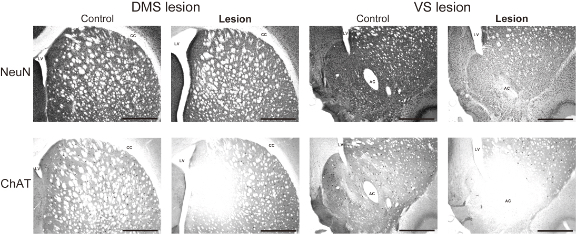
Behavioral flexibility is broadly defined as the ability to change behavioral strategy, according to a change of governing rules. Accumulated evidence suggests the involvement of particular brain areas such as prefrontal cortex and striatum in this function, in which specific brain regions play their own roles. An extension of understanding on neural substrates mediating behavioral flexibility needs a next step beyond the specificity of brain regions: the specific role of different neuronal subtypes. A method utilizing specific neurotoxins enabled us to target and elucidate the role of neurochemically-specific neurons in this ability. In our recent study,[1] we demonstrated a causal role of rat cholinergic interneurons in the striatum in behavioral flexibility, using a new specific immunotoxin targeting neurons containing choline acetyltransferase (ChAT). Comparing non-selective neuronal labeling and specific immunostaining of ChAT neurons indicated that local injections of the immunotoxin successfully and selectively damaged cholinergic neurons (Fig. 1). This result is consistent with a previous study that used Anti-ChAT-SAP to study the medial prefrontal cortex (Cat. #IT-42).[2]
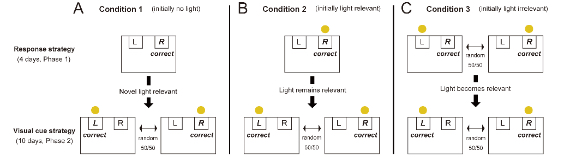
Using the selective lesion, we compared intact rats injected with saline and rats without cholinergic interneurons of either dorsomedial or ventral striatum in a set-shifting task.[3] This task required animals to shift their attention from one stimulus dimension to another to change action strategies, based on a change of behavioral rules. We extended an established task[3] by setting three experimental conditions for a set-shift (Fig. 2), all of which required a change between two strategies involving attention to different stimuli. In all the conditions, animals initially learned to obtain a reward by choosing a Right lever (Fig. 2, Response strategy). Subsequently, after the set-shift, animals faced a change of behavioral rules in which animals had to learn to select a lever indicated by a light cue that randomly illuminated above either lever (Fig. 2, Visual cue strategy). Different manipulations of the light delivery in initial learning made it possible to test different attentional shifts in the next visual cue learning: attention to either 1) a previously absent but now novel light cue (Fig. 2A), 2) a previously relevant and remained relevant cue (Fig. 2B), and a previously irrelevant but now relevant cue (Fig. 2C).
Initial acquisition of response strategy was intact across conditions and treatments, indicating that the striatal cholinergic interneurons are unnecessary for initial learning. By contrast, after a change of behavioral rules occurred, both types of lesions made animals stick to an old strategy. They also showed less exploration for figuring a new rule out. Interestingly, ventral cholinergic ablation disrupted a strategic shift when it required attention to a novel light cue that was introduced as a new important stimulus (Fig. 2A). On the other hand, cholinergic loss in the dorsomedial striatum impaired a set shift when attention to a previously irrelevant cue was needed (Fig. 2C). There was no effect on a shift if the light remained relevant (Fig. 2B). These findings suggest that when facing a change of behavioral rules, striatal cholinergic interneurons play a specific role, namely inhibiting the use of an old strategy and facilitating exploration of a new rule. Furthermore, dorsomedial and ventral striatum cholinergic systems differentially contribute to this function in a highly context-dependent manner. Owing to the prominent targeting method by the Anti-ChAT-SAP, we found a causal role of a neurochemically-specific neuron in behavioral flexibility. This technique is undoubtedly powerful to deepen our knowledge of the causal relationship of particular neuronal types and behavior, and is encouraged for use in studies of different types of behavior.
References: (back to top)
- Aoki S, Liu AW, Zucca A, Zucca S, Wickens JR (2015) Role of Striatal Cholinergic Interneurons in Set-Shifting in the Rat. The Journal of Neuroscience 35:9424-9431.
- Laplante F, Lappi DA, Sullivan RM (2011) Cholinergic depletion in the nucleus accumbens: Effects on amphetamine response and sensorimotor gating. Progress in Neuro-Psychopharmacology and Biological Psychiatry 35:501-509.
- Floresco SB, Block AE, Tse MTL (2008) Inactivation of the medial prefrontal cortex of the rat impairs strategy set-shifting, but not reversal learning, using a novel, automated procedure. Behavioural Brain Research 190:85-96.