Summary of contribution to “Recontres en toxinologie, 2005”
by Ronald G. Wiley, Neurology Service (127) – VA TVHS, 1310 24th Avenue, South, Nashville, TN 37212
The use of targeted toxins in neuroscience research has evolved over the past twenty-plus years from original suicide transport lesions using ricin to highly selective neuron type-specific lesions made with immunotoxins, such as anti-dopamine beta hyroxylase-saporin (anti-DBH-SAP, Cat. #IT-03), and neuropeptide-toxin conjugates, such as substance P-saporin (SP-SAP, Cat. #IT-07).
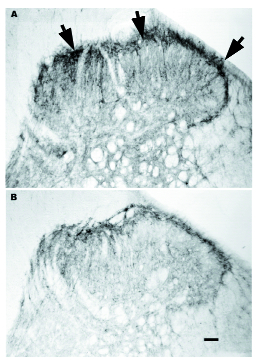
Application of these agents to experiments in the neurobiology of pain began about ten years ago with the development of anti-DBH-SAP which selectively destroys adrenergic and noradrenergic neurons, and SP-SAP which destroys neurons that express neurokinin-1 receptor (NK-1r; Figure 1). Anti-DBH-SAP has been used to show the importance of central noradrenergic neurons in withdrawal from chronic opiate administration and in descending regulation of nociception. Intrathecal injection of SP-SAP produces robust decreases in operant responses to noxious thermal stimuli over a wide range of temperatures with preserved innate reflex nocifensive responses to noxious thermal and mechanical stimuli.[1,2] SP-SAP also profoundly decreases operant hyperalgesia and nocifensive hyperreflexia in a variety of animal models, including topical mustard oil or capsaicin, spinal nerve ligation, carrageenan- and Freund’s adjuvant-induced inflammation.[3,4] SP-SAP reduces responding in the formalin model of persistent pain (phase II). A targeted toxin using a more stable analog of substance P (SSP-SAP, Cat. #IT-11) produces similar effects at lower doses with better specificity.[5]
A similar construct, dermorphin-SAP (Cat. #IT-12), eliminates mu opiate receptor-expressing neurons from the medulla or the substantia gelatinosa of the spinal cord.[6] Medullary dermorphin-SAP injections produce changes in descending regulation of nociception resulting in decreased hyperalgesia and allodynia in a sciatic nerve constriction injury model of neuropathic pain.[7-9] The successes of SP-SAP, SSP-SAP, and dermorphin-SAP suggest a general strategy for targeting neurons expressing specific G-protein coupled receptors. SP-SAP, and perhaps other neuropeptide-toxin conjugates, may have potential in the treatment of chronic intractable pain (see companion article).
Making selective neural lesions has long been an important experimental strategy in neuroscience. The power of this approach depends in large part on the specificity of the lesions. The term “molecular neurosurgery” refers to the use of targeted toxins to produce specific neural lesions based on targeting surface molecules on the neurons of interest.
This work began with suicide transport agents, such as ricin, that were delivered to target neurons by retrograde axonal transport. Suicide transport agents produce anatomically specific lesions but lack cell type selectivity. In order to selectively destroy specific types of neurons, we developed anti-neuronal immunotoxins such as anti-DBH-SAP, anti-SERT-SAP (Cat. #IT-23), and 192-IgG-SAP (Cat. #IT-01). These agents consist of monoclonal antibodies that recognize molecules expressed on the surface of specific types of neurons; the antibody is armed to kill by coupling to the ribosome-inactivating protein, saporin. (The more recent approach to cell type selective lesioning is neuropeptide-toxin conjugates such as SP-SAP and dermorphin-SAP.) Saporin, by itself, normally enters cells very inefficiently, but when coupled to a carrier that induces receptor-mediated endocytosis, saporin uptake can be highly efficient and limited to cells displaying the target molecule. The table below summarizes the targeted toxins available for the study of pain.
Molecular Neurosurgery Agents of Interest in Pain Research
| Agent | Target | Lesioning Use |
| 192 IgG-SAP Cat. #IT-01 | p75NTR, low affinity NGFr (cholinergic basal forebrain, cerebellar Purkingje neurons, postganglionic autonomic neurons, and some primary sensory neurons) | Immunolesioning |
| Anti-DBH-SAP Cat. #IT-03 | Dopamine beta hydroxylase (adrenergic and noradrenergic neurons) | Immunolesioning by application to either dendrites/soma or by retrograde axonal transport |
| Anti-SERT-SAP Cat. #IT-23 | Serotonin transporter (serotonergic neurons) | Immunolesioning |
| Dermorphin-SAP Cat. #IT-12 | Mu opioid receptor (striatal neurons, lamina II dorsal horn nociceptive interneurons, ventromedial medulla) | Direct application, spinal intrathecal injection |
| OX7-SAP Cat. #IT-02 | Thy-1 (all rat neurons and some T lymphocytes) | Suicide transport in both peripheral and central nervous systems |
| SP-SAP and SSP-SAP Cat. #IT-07 and IT-11 | Neurokinin-1 receptor (NK-1r) (striatal cholinergic interneurons, dorsal horn nociceptive projection neurons) | Direct application to dendrites/soma, spinal intrathecal injection |
References: (back to top)
- Khasabov SG, Ghilardi JR, Mantyh PW. J Neurosci 22:9086-9098, 2002.
- Mantyh et al. Science 278:275-279, 1997.
- Nichols et al. Science 286:1558-1561, 1999.
- Vierck CJ, Kline RH, Wiley RG. Neuroscience 119:223-232, 2003.
- Wiley RG, Lappi DA. Neurosci Lett 230:97-100, 1997.
- Wiley RG, Lappi DA. Adv Drug Deliv Rev 55:1043-1054, 2003.
- Burgess et al. J Neurosci 22:5129-5136, 2002.
- Gardell et al. J Neurosci 23:8370-8379, 2003.
- Porreca et al. J Neurosci 21:5281-5288, 2001.
