Contributed by David J Flavell, PhD, The Simon Flavell Leukaemia Research Unit, Division of Cancer Sciences, University of Southampton Medical School, United Kingdom.
I have asked Dr. Flavell to contribute an article on the clinical use of his saporin immunotoxins. Dr. Flavell’s research unit is named after his son, who died from leukemia. At that point, he and his wife, Bee, dedicated their lives to finding a cure for childhood cancers. You are encouraged to contribute to Dr. Flavell’s efforts that are more fully described at the following website:
Douglas A. Lappi, President/Chief Scientific Officer
Immunotoxins have a clinical utility in any human condition where the elimination of an unwanted cell population is desired. The most obvious candidate disease is cancer and consequently the vast majority of early phase clinical studies with immunotoxins have centered on the treatment of human malignancies with the goal of eliminating cancer cells from the patient’s body. Most of these clinical trials have been conducted using ricin A chain or blocked ricin1 as the toxin component. Whilst there have been anti-tumor responses with ricin-based immunotoxins, particularly in hematological malignancies,2 their toxicity profile has limited their clinical usefulness and interest in their utility has consequently dropped away. In contrast, there have been very few clinical studies with saporin-based immunotoxins (Fig 1). The earliest study was conducted by Falini and his associates3 in Italy and Germany utilizing a CD30 antibody coupled to saporin for the treatment of four patients with advanced Hodgkin’s disease. Two patients showed dramatic responses to treatment and no serious side effects were observed. In particular, vascular leak syndrome, a potentially fatal toxicity that had been associated with ricin-based immunotoxins, was not observed.
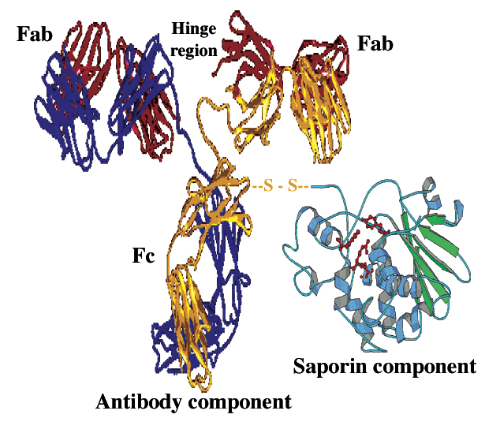
These initial results encouraged us to undertake preclinical investigations with immunotoxins directed against the B-cell restricted CD19 antigen for B-lymphoid tumors and against the CD38 molecule for multiple myeloma. The development of such therapeutic molecules required robust preclinical investigations to determine their toxicity profiles in animals, the normal tissue-binding specificities, and to demonstrate their selective anti-tumor activity both in vitro and in vivo. Such preclinical investigations are mandatory to justify taking an experimental drug into patients and to ensure, as far as possible, safety of the drug. A poignant reminder here was the disastrous peripheral nerve damage experienced by women with breast cancer treated with a ricin A chain-based immunotoxin which unexpectedly also targeted nervous tissue.4 The most sensitive in vitro method for determining the selective cytotoxic potency of saporin-based immunotoxins is by measuring their ability to selectively inhibit protein synthesis in antigen-expressing cell lines in a dose-dependent manner. This is clearly shown in Figure 2, where increasing concentrations of two immunotoxins: anti-CD19 (BU12-Saporin) and anti-CD38 (OKT10-SAP) show decreasing protein synthesis levels in a target CD19+ CD38+ pediatric acute lymphoblastic leukaemia (ALL) cell line. Note that when the two immunotoxins were used in combination no improvement in performance was seen. When the same two immunotoxins were used individually in vivo in SCID mice xenografted with the same ALL cell line, they exerted therapeutic activity in line with the data obtained from the in vitro study. However, when the two immunotoxins were used in combination the therapeutic effect was significantly better than either individual drug used alone (Fig 3). This in vivo result could not have been predicted from the short-term protein synthesis inhibition assay. We have also made similar findings with in vivo models of human B-cell lymphoma5 and T-cell leukaemia.6 These observations have provided us with the longer-term goal of conducting clinical trials with combinations of different immunotoxins.
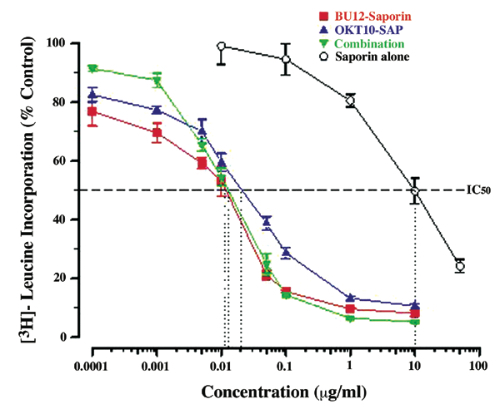
Figure 2 Protein synthesis inhibition in the CD19+/CD38+ acute lymphoblastic cell line, NALM-6, following 48h exposure to increasing concentrations of BU12-Saporin, OKT10-SAP, a combination of both immunotoxins, or saporin alone. The IC50 is shown as the point on the x-axis intercept representing the concentration of ITthat inhibits protein synthesis in the target cell line by 50% relative to untreated control cells. Note that the use of a combination of both ITs does not result in any increased cytotoxicity for the target cell line in this particular assay.
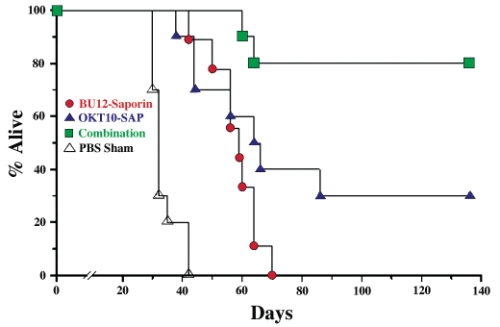
Following preclinical evaluation my laboratory is now directly involved in two separate Phase I clinical trials with BU12-Saporin and OKT10-SAP currently underway in the United Kingdom. Working together with the United Kingdom Children’s Cancer Study Group and Cancer Research UK, we are now treating children with relapsed ALL with BU12-Saporin at 11 children’s cancer units and adult myeloma patients with OKT10-SAP at two centers. These are early phase dose escalation studies whose primary aims are to determine the maximum tolerated dose (MTD) and dose limiting toxicities (DLT) for both saporin immunoconjugates for use in subsequent Phase II studies. The secondary objective of these studies is to define the pharmacokinetic profile of these drugs in patients and to document any tumor responses.
In conventional Phase I studies, cohorts of three patients receive a fixed dose of drug and are then monitored for toxic side effects, which are carefully graded according to strictly laid down criteria. If toxic side effects occur below grade 3, then the next cohort of patients is permitted to receive the next dose level up. The trial continues in this way until the MTD is reached. In our ongoing studies with OKT10-SAP in adult myeloma patients a dose level of 40 μg/kg/day x 5 days has been reached so far without any serious drug-related side effects. Encouragingly, we have not encountered vascular leak syndrome in any patient. Approximately half of the patients have developed human anti-mouse antibodies (HAMA) and a similar number human anti-saporin antibodies (HASA). Pharmacokinetic analysis, though only partially completed and not yet formally analyzed, has revealed some major differences in peak levels and decay rates between different patients. This probably reflects differences in tumor burden between individual patients— it is predicted that patients with higher tumor burdens will achieve lower peak serum levels and a shorter drug half life due to rapid removal of drug from the circulation by the larger antigenic “sink” that a larger tumor burden provides. The BU12- Saporin study in relapsed pediatric ALL is at a less advanced stage and has so far recruited six patients. Once the MTD has been established for both drugs then we can move on to Phase II studies whose primary objective will be to determine the response rate. Then the really exciting work begins!
References
- Vitetta ES, Thorpe PE, Uhr JW (1993) Immunotoxins: Magic Bullets or Misguided Missiles? Immunol Today 14:252-259.
- Vitetta ES, Stone M, Amlot P et al. (1991) Phase I immunotoxin trial in patients with B-cell lymphoma. Cancer Res 51:4052-4058.
- Falini B, Bolognesi A, Flenghi L et al. (1992) Response of refractory Hodgkin’s disease to monoclonal anti-CD30 immunotoxin. Lancet 339:1195-1196.
- Gould BJ, Borowitz MJ, Groves ES et al. (1989) Phase I study of an anti-breast cancer immunotoxin by continuous infusion: Report of a targeted toxic effect not predicted by animal studies. J Natl Cancer Inst 81(10):775-781.
- Flavell DJ, Noss A, Pulford KA, Ling N, Flavell SU (1997) Systemic therapy with 3BIT, a triple combination cocktail of anti-CD19, -CD22, and -CD38-saporin immunotoxins, is curative of human B-cell lymphoma in severe combined immunodeficient mice. Cancer Res 57(21):4824- 4829.
- Flavell DJ, Boehm DA, Noss A, Warnes SL, Flavell SU (2001) Therapy of human T-cell acute lymphoblastic leukaemia with a combination of anti-CD7 and anti-CD38-SAPORIN immunotoxins is significantly better than therapy with each individual immunotoxin. Br J Cancer 84(4):571- 578.
- Messmann RA, Vitetta ES, Headlee D et al. (2000) A phase I study of combination therapy with immunotoxins IgG-HD37-deglycosylated ricin A chain (dgA) and IgG-RFB4-dgA(Combotox) in patients with refractory CD19(+), CD22(+) B cell lymphoma. 6(4):1302-1313.
