Early last month (March 2001), Advanced Targeting Systems (ATS) received funding from the National Institute of Mental Health (NIMH) to begin toxicology/safety studies of Substance P-Saporin (SP-SAP), a potential therapeutic for the treatment of chronic pain. The studies will be completed under the direction of three scientists who are experts in their respective fields.
Dr. Douglas Lappi (President and Chief Scientific Officer of ATS) is principal investigator for the project and will oversee the various aspects of the studies. He is an expert in the design, construction and testing of targeted toxins. His laboratory will be producing the drug and performing quality control assays throughout the project.
Dr. Tony Yaksh (Professor of Anesthesiology and Pharmacology at the University of California, San Diego School of Medicine) will direct the administration of the drug. He is the leading expert in spinal cord delivery of experimental agents. The dog is one of the species routinely used to satisfy most regulatory requirements for drug safety evaluation. The studies will assess safety from four points: 1) intrathecal dose ranging to determine the maximum tolerated dose, 2) kinetics of cerebral spinal fluid to determine how the drug penetrates spinal tissue, is redistributed and eliminated, 3) histopathology to determine impact of drug on organs and tissue, and 4) spinal GLP safety studies to determine physiological (heart and respiratory rate, blood pressure) and behavioral (arousal, muscle tone, coordination) impacts of drug administration (4).
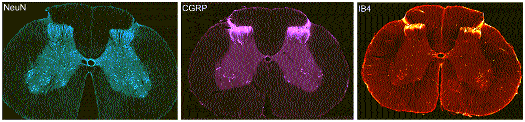
Dr. Patrick Mantyh (Professor, University of Minnesota, Minneapolis) has established the efficacy of SP-SAP in rats and is internationally acclaimed for his immunohistochemical analysis. His laboratory will measure parameters involving the efficacy and specificity of the SP-SAP treatment. Immunohistochemistry will help in determining where the drug travels and what impact, if any, it has on spinal cord neurons (See Figure).
Figure Legend: This figure shows the staining of NeuN, CGRP and IB4 in the canine spinal cord. Immunostaining is one of the tests that will be used for determination of specificity and evaluation of bystander effects by SP-SAP. Confocal photomicrographs show the pattern of immunohistochemical labeling of the neuronal nuclear marker, NeuN, a peptidergic sensory nerve fiber marker, CGRP, and the non-peptidergic sensory nerve fiber marker, IB4, in the dog spinal cord. The NeuN staining is distributed throughout the entire gray matter, while the staining for the sensory fibers (CGRP, IB4) is localized to the dorsal horn (Photo supplied by Dr. Patrick Mantyh).
The development of SP-SAP was first published in 1997 (1) by Drs. Ronald G. Wiley and Douglas Lappi, two of the founders of ATS. Their collaboration with Dr. Patrick Mantyh led to two publications in the journal Science (2, 3). These three articles describe the results of experiments with SP-SAP in the rat.
SP-SAP is a targeted toxin that permanently eliminates cells that bear the Substance P receptor. This receptor is one of many involved in the transmission of pain signals to the brain. There are two general categories of pain to be considered in this process: 1) acute pain, a physiologically important survival tool (rose thorn pricking finger, cat claws scratching cheek), and 2) chronic or noxious pain (pain that persists beyond normal healing time), often the cause of severe pathological states. The scientists used standard models of chronic pain to determine that the perception of noxious pain in the models was greatly reduced in those animals who received injections of SP-SAP. But probably just as important, the perception of acute pain was left intact. This was an extraordinarily important finding and led ATS to the decision to begin the process of developing SP-SAP as a drug.
Over the next few months, ATS will be interacting with the Center for Drug Evaluation and Research. This organization is part of the U.S. Food and Drug Administration and will evaluate the drug development plan and make determinations about the composition and guidelines for the initial clinical trials in humans. Their preliminary feedback has been a recommendation to begin clinical trials in patients with terminal cancer whose pain is no longer treatable with opioid-based drugs such as morphine. The size of this patient population may qualify SP-SAP for development as an orphan drug.
ATS is optimistic about the therapeutic possibilities of SP-SAP. The funding from NIMH is an important first step in getting the drug development process under way. The process has already begun to obtain the additional funding needed to complete the toxicology tests required by the FDA. The present goal is to be able to begin the first clinical trials in humans before the end of 2002. Progress reports will be printed in our newsletter and on the ATS website.
References
- Wiley RG, Lappi DA (1997) Destruction of neurokinin-1 receptor expressing cells in vitro and in vivo using substance P-saporin. Neurosci Lett 230:97-100.
- Mantyh PW, Rogers S, Honore P, Allen B, Ghilardi JR, Li J, Daughters RS, Vigna SR, Lappi DA, Wiley RG, Simone DA (1997) Inhibition of hyperalgesia by ablation of lamina I spinal neurons expressing the substance P receptor. Science 278:275-279.
- Nichols ML, Allen BJ, Rogers SD, Ghilardi JR, Honore P, Li J, Lappi DA, Simone DA, Mantyh PW (1999) Transmission of chronic nociception by spinal neurons expressing the substance P receptor. Science 286:1558-1561.
- Yaksh TL, Rathbun ML, Provencher JC. (1999) Preclinical safety evaluation for spinal drugs. In: Spinal Drug Delivery, Yaksh TL (ed.), Elsevier Science B.V., Amsterdam, pp. 417-437.