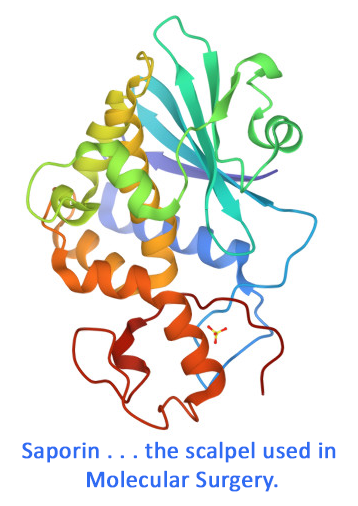
Camelids produce functional IgG isotypes that do not incorporate light chains. Comparative studies of old world camelids (Camelus bactrianus and Camelus dromedarius) and new world camelids (Lama pacos, Lama glama and Lama vicugna) have shown that heavy-chain-only immunoglobulins represent between 35% – 70% of total IgG in the sera of all species. At present, three subclasses of camelid IgG have been identified (IgG1, IgG2, IgG3), of which IgG2 and IgG3 lack the light chains. IgG2 consists of dimers of short heavy chains, which are characterized by a normal Fc region without a CH1 domain. IgG2 shows a molecular weight of approximately 90 kDa and this smaller size improves its bio-distribution and allows a better tissue penetration. Of special importance is the long complementary determining region (CDR) loop which inserts deep into the active site of an enzyme enabling a complete neutralization of an enzyme. Anti-Llama IgG2 generated in rabbit detects specifically Llama IgG2 isotype. This anti-Llama secondary Antibody is suitable for western blot, ELISA, ChIP and immunohistochemistry as well as other more general immunoassays.